Advertisements
Advertisements
प्रश्न
Give the trends in atomic size on moving across the period left to right.
उत्तर
Atomic size decreases from left to right as the Atomic Number increases by one at each stage, with one proton added to the nucleus and one electron added to the valence shell. As nuclear attraction rises, the size of the atom decreases.
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Which is greater in size an atom or an anion?
Write the name and symbol of the element from the description.
The atom having the smallest size.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
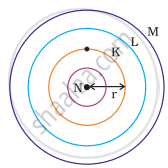
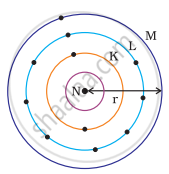
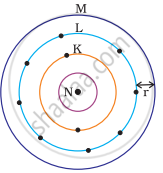
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)