Advertisements
Advertisements
प्रश्न
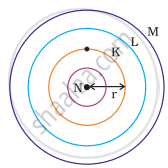
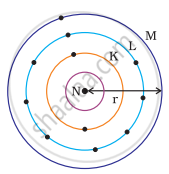
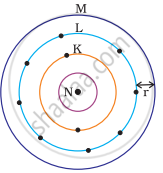
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
विकल्प
(i) and (ii)
(ii) and (iii)
(iii) and (iv)
(i) and (iv)
उत्तर
(ii) and (iii)
Explanation -
The atomic radius(r) of an atom is the distance between the centre of the nucleus and the outermost shell of an isolated atom. In representation (ii), the outermost shell is K and atomic radius is correctly depicted from centre of nucleus to shell K while in representation (iii), the outermost shell is M and atomic radius is correctly depicted from centre of nucleus to shell M.
APPEARS IN
संबंधित प्रश्न
What do you understand by atomic size? State its unit?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Write scientific reason.
Atomic radius goes on increasing down a group.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
On moving from left to right in a periodic table, the size of the atom _______.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.