Advertisements
Advertisements
प्रश्न
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
उत्तर
The element with largest atomic size: Lithium (Li)
Notes
Concept Insight: Lithium has the largest atomic size since it is an alkali metal i.e., belongs to group 1 and we know that as we move from left to right in a period atomic size decreases. So lithium has largest size while fluorine has smallest size in second period.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
The size of an atom is indicated by its _______.
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
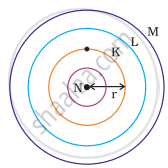
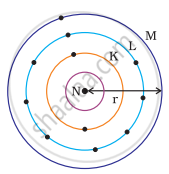
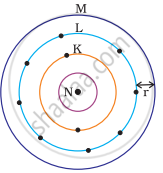
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.