Advertisements
Advertisements
Question
Moving from left to right, the size of the atom decreases.
Options
Right
Wrong
Solution
Moving from left to right, the size of the atom decreases- Right
APPEARS IN
RELATED QUESTIONS
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
What do you understand by atomic size? State its unit?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
Which of the following is the correct order of size?
Which among the following elements has the largest atomic radii?
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I
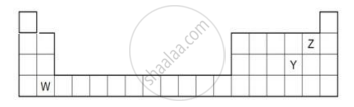
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
An element with the largest atomic radius among the following is ______.
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Which one of the following has the largest atomic radius?
Which one has the largest size?
