Advertisements
Advertisements
Question
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Solution
Beryllium > Carbon > Fluorine
Explanation:
As we proceed along a period, the size of the atom decreases due to increased nuclear pull.
APPEARS IN
RELATED QUESTIONS
Which is greater in size an atom or a cation?
Write scientific reason.
Atomic radius goes on increasing down a group.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
The size of an atom is indicated by its _______.
Atomic radius is expressed in the unit _______.
Which of the following is the correct order of atomic size?
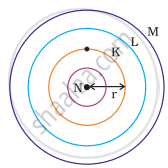
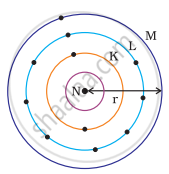
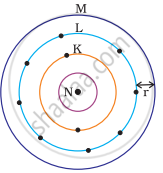
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.