Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
उत्तर
Beryllium > Carbon > Fluorine
Explanation:
As we proceed along a period, the size of the atom decreases due to increased nuclear pull.
APPEARS IN
संबंधित प्रश्न
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Nanometer unit is used to measure atomic radius.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
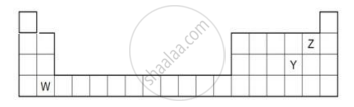
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
Which one has the largest size?
