Advertisements
Advertisements
प्रश्न
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

उत्तर
- H, He, Li, Be, B, C, N, O, F, Ne, Na, Mg, Al, Si, P, S, Cl, Ar, K, Ca
- Group 1 — H, Li, Na, K
Group 2 — Be, Mg, Ca
Group 13 — B, Al
Group 14 — C, Si
Group 15 — N, P
Group 16 — O, S
Group 17 — F, Cl
Group 18 — He, Ne, Ar
APPEARS IN
संबंधित प्रश्न
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Arrange the following in order of increasing radii:
CI- , CI
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
On moving from left to right in a periodic table, the size of the atom _______.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Which of the following is the correct order of size?
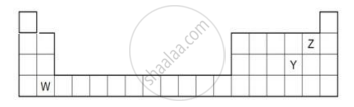
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
