Advertisements
Advertisements
Question
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

Answer in Brief
Solution
- H, He, Li, Be, B, C, N, O, F, Ne, Na, Mg, Al, Si, P, S, Cl, Ar, K, Ca
- Group 1 — H, Li, Na, K
Group 2 — Be, Mg, Ca
Group 13 — B, Al
Group 14 — C, Si
Group 15 — N, P
Group 16 — O, S
Group 17 — F, Cl
Group 18 — He, Ne, Ar
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Why is the size of sodium is greater than magnesium?
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
The size of an atom is indicated by its _______.
Atomic radius is expressed in the unit _______.
Moving from left to right, the size of the atom decreases.
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
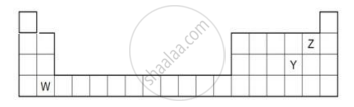
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
