Advertisements
Advertisements
प्रश्न
Complete and balance the following equation:

उत्तर
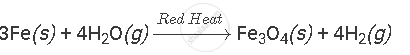
APPEARS IN
संबंधित प्रश्न
.................... liberated when acetic acid reacts with sodium metal.
(a) Hydrogen
(b) Chlorine
(c) Oxygen
(d) Nitrogen
Write the electron-dot structures for sodium, oxygen and magnesium.
Write equation for the reaction of sodium with oxygen.
Which of the following elements would yield a neutral oxide?
Na, S, C, K, H
What is the action of water on magnesium ? Write equation of the chemical reaction involved.
The metals which can produce amphoteric oxides are:
(a) sodium and aluminium
(b) zinc and potassium
(c) calcium and sodium
(d) aluminium and zinc
Which one of the following four metals would be displaced from the solution of its salt by the other three metals?
(a) Zn
(b) Ag
(c) Cu
(d) Mg
Explain the following:
Reactivity of aluminium decrease if it is dipped in nitric acid.
Arrange the following metals in the increasing order of their reactivities:
Copper, Zinc, Aluminium, and Iron
An element X is soft and can be cut with a knife. This is very reactive to air and cannot be kept open in the air. It reacts vigorously with water. Identify the element from the following.
