Advertisements
Advertisements
प्रश्न
Complete the following reactions
NH3+3Cl2(excess) ---->
उत्तर
Complete chemical reactions are:
NH3 + 3 Cl2 (excess) → NCl3 + 3 HCl
APPEARS IN
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Write IUPAC names of the following compounds
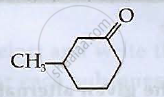
Write the IUPAC names of the following coordination compounds:
`[PtCl_2(NH_3)_4][PtCl_4]`
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The coordination number of the central ion may be obtained from:
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
The complex which has no d electrons in the central atom is:-
Glycinato ligand is ______.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
