Advertisements
Advertisements
प्रश्न
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
विकल्प
Assertion and reason both are true, reason is correct explanation of assertion.
Assertion and reason both are true but reason is not the correct explanation of assertion.
Assertion is true, reason is false.
Assertion is false, reason is true.
उत्तर
Assertion and reason both are true but reason is not the correct explanation of assertion.
Explanation:
In the complexes, \[\ce{Co}\] exists as \[\ce{Co^{2+}}\] and \[\ce{Fe}\] as \[\ce{Fe^{2+}}\]. Both of the complexes becomes stable by oxidation of metal ion to \[\ce{Co^{3+}}\] and \[\ce{Fe^{3+}}\].
APPEARS IN
संबंधित प्रश्न
Write structures of compounds A, B and C in of the following reactions
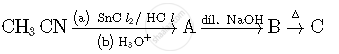
Complete the following reactions
NH3+3Cl2(excess) ---->
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Ethylene diamine tetraacetate (EDTA) ion is ______.
What are Homoleptic complexes?
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
Give two examples of didentate ligand.
