Advertisements
Advertisements
प्रश्न
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
पर्याय
Assertion and reason both are true, reason is correct explanation of assertion.
Assertion and reason both are true but reason is not the correct explanation of assertion.
Assertion is true, reason is false.
Assertion is false, reason is true.
उत्तर
Assertion and reason both are true but reason is not the correct explanation of assertion.
Explanation:
In the complexes, \[\ce{Co}\] exists as \[\ce{Co^{2+}}\] and \[\ce{Fe}\] as \[\ce{Fe^{2+}}\]. Both of the complexes becomes stable by oxidation of metal ion to \[\ce{Co^{3+}}\] and \[\ce{Fe^{3+}}\].
APPEARS IN
संबंधित प्रश्न
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
Write applications of co-ordination compounds in medicine and electroplating.
Write structures of compounds A, B and C in of the following reactions
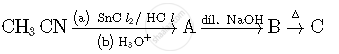
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
The most stable ion is:-
What are Homoleptic complexes?
