Advertisements
Advertisements
प्रश्न
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
उत्तर
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Explanation:
Complexes in which a metal is bound to more than one kind of donor groups are known as heteroleptic. This condition is satisfied by complexes mentioned at \[\ce{[Cr(NH3)6]^{3+}}\] and \[\ce{[Co(NH3)4]Cl2]}\] where the donor groups are \[\ce{NH3}\] and \[\ce{Cl2}\].
APPEARS IN
संबंधित प्रश्न
Explain the following, giving two examples:
Coordination entity
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
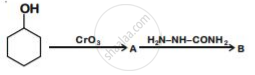
Write structures of compounds A and B of the following reaction :
The coordination number of the central ion may be obtained from:
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Oxidation number of cobalt in K[Co(CO)4] is
The nature of hybridisation in the ammonia molecule is
Glycinato ligand is ______.
Which of the following species cannot act as a ligand? Give reason.
Give two examples of ambidentate ligand.
