Advertisements
Advertisements
Question
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Solution
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Explanation:
Complexes in which a metal is bound to more than one kind of donor groups are known as heteroleptic. This condition is satisfied by complexes mentioned at \[\ce{[Cr(NH3)6]^{3+}}\] and \[\ce{[Co(NH3)4]Cl2]}\] where the donor groups are \[\ce{NH3}\] and \[\ce{Cl2}\].
APPEARS IN
RELATED QUESTIONS
Write structures of compounds A, B and C in of the following reactions
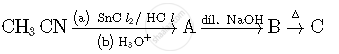
Write the structures of compounds A, B and C in the following reactions:
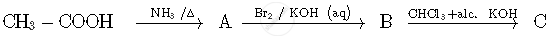
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
The coordination number of the central ion may be obtained from:
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
Ethylene diaminetetraacetate (EDTA) ion is ______
What are Heteroleptic complexes?
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Explain the following, giving two examples:
Coordination polyhedron
