Advertisements
Advertisements
Question
Write the structures of compounds A, B and C in the following reactions:
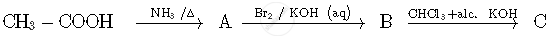
Solution
A: CH3CONH2 (Ethanamide)
B: CH3NH2 (Methanamine)
C: CH3NC (Methyl isocyanide)
APPEARS IN
RELATED QUESTIONS
Write structures of compounds A, B and C in of the following reactions
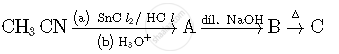
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
In which of the following compounds, the oxidation number of iodine is fractional?
Glycinato ligand is ______.
What is a chelate complex? Give one example.
Explain the following, giving two examples:
Coordination polyhedron
What is meant by ambidentate ligand?
