Advertisements
Advertisements
प्रश्न
Write the structures of compounds A, B and C in the following reactions:
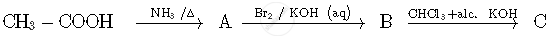
उत्तर
A: CH3CONH2 (Ethanamide)
B: CH3NH2 (Methanamine)
C: CH3NC (Methyl isocyanide)
APPEARS IN
संबंधित प्रश्न
Write applications of co-ordination compounds in medicine and electroplating.
The ligand triethylenetetramine is _______.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
The nature of hybridisation in the ammonia molecule is
Which one of the following ligands forms a chelate?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
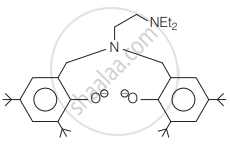
The following ligand is:
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
