Advertisements
Advertisements
प्रश्न
The ligand triethylenetetramine is _______.
पर्याय
monodentate
bidentate
tridentate
tetradentate
उत्तर
The ligand triethylenetetramine is tetradentate.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
What is meant by unidentate ligand?
Write the structures of compounds A, B and C in the following reactions
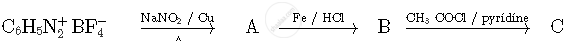
Write the IUPAC names of the following coordination compounds:
`[PtCl_2(NH_3)_4][PtCl_4]`
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Which one of the following does not achieve an octet of electrons in the central atom?
Oxidation number of cobalt in K[Co(CO)4] is
In which of the following compounds, the oxidation number of iodine is fractional?
Which one of the following ligands forms a chelate?
The complex which has no d electrons in the central atom is:-
According to IUPAC nomenclatures, sodium nitroprusside is named as
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Ethylene diaminetetraacetate (EDTA) ion is ______
Why chelate complexes are more stable than complexes with unidentate ligands?
The following ligand is:
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Glycinato ligand is ______.
What is a chelate complex? Give one example.
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Ligand
What is meant by didentate ligand?
