Advertisements
Advertisements
प्रश्न
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
उत्तर
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
Explanation:
Complexes in which a metal is bound to only one kind of donor groups, e.g., \[\ce{[Co(NH3)6]^{3+}}\], are known as homoleptic.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Write the IUPAC names of the following coordination compounds:
`[PtCl_2(NH_3)_4][PtCl_4]`
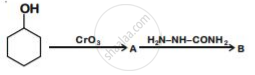
Write structures of compounds A and B of the following reaction :
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The coordination number of the central ion may be obtained from:
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
In which of the following compounds, the oxidation number of iodine is fractional?
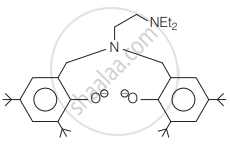
The following ligand is:
Explain the following, giving two examples:
Coordination number
Give two examples of ambidentate ligand.
