Advertisements
Advertisements
प्रश्न
Explain the following, giving two examples:
Coordination number
उत्तर
The coordination number (CN) of a metal ion in a complex can be defined as the number of ligand donor atoms to which the metal is directly bonded. For example, in the complex ions, [PtCl6]2− and [Ni(NH3)4]2+, the coordination numbers of Pt and Ni are 6 and 4, respectively. Similarly, in the complex ions, [Fe(C2O4)3]3− and [Co(en)3]3+, the coordination number of both Fe and Co is 6 because \[\ce{C2O^{2−}4}\] and en (ethane-1, 2-diamine) are didentate ligands.
संबंधित प्रश्न
Amongst the following, the most stable complex is:
Write IUPAC names of the following compounds:
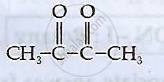
Write structures of compounds A and B of the following reaction :

A group of atoms can function as a ligand only when:
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
According to IUPAC nomenclatures, sodium nitroprusside is named as
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Ligand
