Advertisements
Advertisements
प्रश्न
Amongst the following, the most stable complex is:
पर्याय
[Fe(H2O)6]3+
[Fe(NH3)6]3+
[Fe(C2O4)3]3−
[FeCl6]3−
उत्तर
[Fe(C2O4)3]3−
Explanation:
\[\ce{C2O^{2-}_4}\] is a bidentate ligand, it forms the most stable complex.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
Write applications of co-ordination compounds in medicine and electroplating.
Write the structures of compounds A, B and C in the following reactions
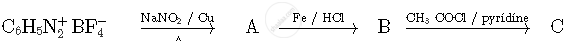
Write the IUPAC names of the following coordination compounds:
`[PtCl_2(NH_3)_4][PtCl_4]`
The ligand triethylenetetramine is _______.
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
The coordination number of the central ion may be obtained from:
In which of the following compounds the oxidation state of the nickel atom is 0?
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Oxidation number of carbon in CH2Cl2 is
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Glycinato ligand is ______.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
