Advertisements
Advertisements
प्रश्न
Amongst the following, the most stable complex is:
विकल्प
[Fe(H2O)6]3+
[Fe(NH3)6]3+
[Fe(C2O4)3]3−
[FeCl6]3−
उत्तर
[Fe(C2O4)3]3−
Explanation:
\[\ce{C2O^{2-}_4}\] is a bidentate ligand, it forms the most stable complex.
APPEARS IN
संबंधित प्रश्न
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
What happens when PCl5 is heated? Write the equations involved.
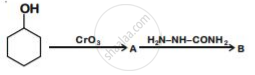
Write structures of compounds A and B of the following reaction :
The coordination number of the central ion may be obtained from:
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Which one of the following does not achieve an octet of electrons in the central atom?
Oxidation number of cobalt in K[Co(CO)4] is
Which one of the following ligands forms a chelate?
According to IUPAC nomenclatures, sodium nitroprusside is named as
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
Give two examples of unidentate ligand.
What is meant by ambidentate ligand?
