Advertisements
Advertisements
प्रश्न
Give two examples of unidentate ligand.
उत्तर
- Cl−
- H2O
- NH3
APPEARS IN
संबंधित प्रश्न
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Write IUPAC names of the following compounds:
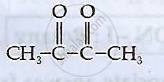
Write structures of compounds A, B and C in of the following reactions
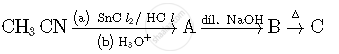
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the structures of compounds A, B and C in the following reactions:
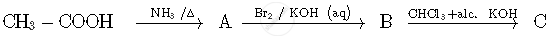
Write the IUPAC names of the following coordination compounds:
`[PtCl_2(NH_3)_4][PtCl_4]`
The ligand triethylenetetramine is _______.
A group of atoms can function as a ligand only when:
Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Which one of the following does not achieve an octet of electrons in the central atom?
Oxidation number of cobalt in K[Co(CO)4] is
Oxidation number of carbon in CH2Cl2 is
Which one of the following ligands forms a chelate?
According to IUPAC nomenclatures, sodium nitroprusside is named as
What are Homoleptic complexes?
Which of the following species cannot act as a ligand? Give reason.
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
Explain the following, giving two examples:
Heteroleptic
