Advertisements
Advertisements
प्रश्न
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
विकल्प
\[\ce{Cu^{2+} + 4NH3 ⇌ [Cu(NH3)4]^{2+}, logK = 11.6}\]
\[\ce{Cu^{2+} + 4CN^- ⇌ [Cu(CN)4]^{2-}, logK = 27.3}\]
\[\ce{Cu^{2+} + 2en ⇌ [Cu(en)2]^{2+}, logK = 15.4}\]
\[\ce{Cu^{2+} + 4H2O ⇌ [Cu(H2O)4]^{2+}, logK = 8.9}\]
उत्तर
\[\ce{Cu^{2+} + 4CN^- ⇌ [Cu(CN)4]^{2-}, logK = 27.3}\]
Explanation:
The greater the value of log K, the greater will be stability of complex compound formed.
For reaction,
\[\ce{Cu^{2+} + 4CN^{-} -> [Cu(CN)4]^{2-}}\]
`"K" = (["Cu"(("CN")_4)^(2-)])/(["Cu"^(2+)]["CN"^(-)]^4)` and log K = 27.3
For this reaction, log K has highest value among the given four reactions. Hence, K will also be higher among these four, i.e., stability of the complexes will be highest among these four complexes.
APPEARS IN
संबंधित प्रश्न
What is meant by unidentate ligand?
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
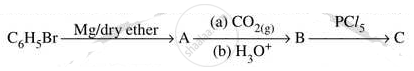
Write structures of compounds A, B and C in of the following reactions
Complete the following reactions
NH3+3Cl2(excess) ---->
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
According to IUPAC nomenclatures, sodium nitroprusside is named as
The following ligand is: