Advertisements
Advertisements
प्रश्न
What is meant by unidentate ligand?
उत्तर
When a ligand is bound to a metal ion through a single donor atom, the ligand is called unidentate.
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
IUPAC name of the following compound is

(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC names of the following compounds:
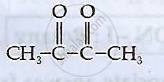
Write structures of compounds A, B and C in of the following reactions
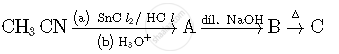
Which of the following represents a chelate ligand?
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
In which of the following compounds the oxidation state of the nickel atom is 0?
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Oxidation number of carbon in CH2Cl2 is
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The most stable ion is:-
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
What is meant by the chelate effect? Give an example.
What is meant by didentate ligand?
