Advertisements
Advertisements
प्रश्न
IUPAC name of the following compound is

(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
उत्तर
3-Bromo-3,4-dimethylheptane
APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Amongst the following, the most stable complex is:
Write structures of compounds A, B and C in of the following reactions
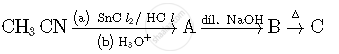
Write structures of compounds A and B of the following reaction :

The oxidation number of Fe in K4[Fe(CN)6] is ____________.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
In which of the following compounds, the oxidation number of iodine is fractional?
Which of the following is an ionic ligand?
Ethylene diaminetetraacetate (EDTA) ion is ______
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
The one that will show optical activity is: (en = ethane 1, 2-diamine)
What is a chelate complex? Give one example.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Ligand
Explain the following, giving two examples:
Homoleptic
What is meant by didentate ligand?
Give two examples of ambidentate ligand.
