Advertisements
Advertisements
प्रश्न
What is a chelate complex? Give one example.
उत्तर
When a di- or polydentate ligand uses its two or more donor atoms simultaneously to bind a single metal ion, it is said to be a chelate ligand. Such complexes are called chelate complexes.
For example: [CoCl2(en)2]+
APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write structures of compounds A and B of the following reaction :
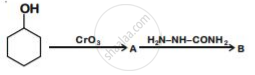
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
Which one of the following does not achieve an octet of electrons in the central atom?
The nature of hybridisation in the ammonia molecule is
What is meant by the chelate effect? Give an example.
