Advertisements
Advertisements
प्रश्न
उत्तर
Structure of the given compounds are:

1) As we can see in the above figures, (A), contains the least steric hindrance so towards the SN2 reaction 1- bromopentane will be most reactive.
2) 2-bromopentane (Figure B) has chiral carbon in it. So, this compound is optically active.
3) Towards the β-elimination 2-bromo-2-methylbutane will be most reactive. Since it will form most stable alkene (on account of highest no. of α-hydrogens).
APPEARS IN
संबंधित प्रश्न
Amongst the following, the most stable complex is:
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
The ligand triethylenetetramine is _______.
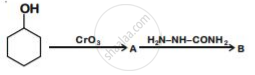
Write structures of compounds A and B of the following reaction :
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
What are Homoleptic complexes?
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
The one that will show optical activity is: (en = ethane 1, 2-diamine)
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
