Advertisements
Advertisements
प्रश्न
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
उत्तर
In an aqueous solution of the complex, 3 ions are produced.
\[\ce{[Co(NH3)6]Cl2 <=> [Co(NH3)6]^{2+} + 2Cl^-}\]
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
How is Benzonitrile converted to Benzophenone?
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
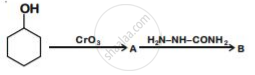
Write structures of compounds A and B of the following reaction :
The coordination number of the central ion may be obtained from:
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
The nature of hybridisation in the ammonia molecule is
