Advertisements
Advertisements
प्रश्न
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
उत्तर
If it forms silver chloride then there is one free chlorine atom outside the coordination sphere. The structural formula has to be \[\ce{[Cr(H2O)4Cl2]Cl}\]. The name of this complex is tetraaquadichlorido chromium (III) chloride.
APPEARS IN
संबंधित प्रश्न
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Amongst the following, the most stable complex is:
Write structures of compounds A, B and C in of the following reactions
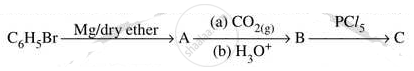
Complete the following reactions
NH3+3Cl2(excess) ---->
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
What is meant by the chelate effect? Give an example.
What is meant by didentate ligand?
