Advertisements
Advertisements
प्रश्न
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
उत्तर
If it forms silver chloride then there is one free chlorine atom outside the coordination sphere. The structural formula has to be \[\ce{[Cr(H2O)4Cl2]Cl}\]. The name of this complex is tetraaquadichlorido chromium (III) chloride.
APPEARS IN
संबंधित प्रश्न
Explain the following, giving two examples:
Coordination entity
Write IUPAC names of the following compounds:
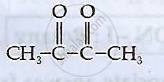
Write applications of co-ordination compounds in medicine and electroplating.
Write the IUPAC name of the following complex : [Co(NH3)5(CO3)]Cl.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Which of the following is an ionic ligand?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Glycinato ligand is ______.
Explain the following, giving two examples:
Heteroleptic
