Advertisements
Advertisements
प्रश्न
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
उत्तर
An optically active \[\ce{[M(AA)2X2]^{n+}}\] indicates civ-octahedral structure e.g. \[\ce{cis-[Pt(en)2Cl2]^{2+}}\] or \[\ce{cis-[Cr(en)2Cl2]^+}\]
APPEARS IN
संबंधित प्रश्न
Write structures of compounds A, B and C in of the following reactions
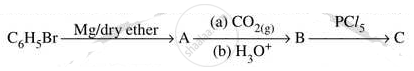
Write structures of compounds A, B and C in of the following reactions
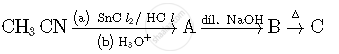
Complete the following reactions
NH3+3Cl2(excess) ---->
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
Oxidation number of carbon in CH2Cl2 is
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
Which of the following ligands can exhibit linkage isomerism?
Ethylene diamine tetraacetate (EDTA) ion is ______.
What is meant by ambidentate ligand?
Give two examples of ambidentate ligand.
