Advertisements
Advertisements
प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
उत्तर
APPEARS IN
संबंधित प्रश्न
Write IUPAC names of the following compounds:
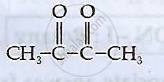
What happens when PCl5 is heated? Write the equations involved.
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the structures of compounds A, B and C in the following reactions
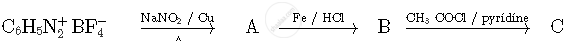
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
In which of the following compounds the oxidation state of the nickel atom is 0?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
What are Heteroleptic complexes?
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
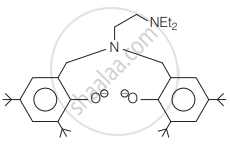
The following ligand is:
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Explain the following, giving two examples:
Coordination polyhedron
Explain the following, giving two examples:
Coordination number
Give two examples of unidentate ligand.
What is meant by didentate ligand?
Give two examples of ambidentate ligand.
