Advertisements
Advertisements
प्रश्न
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
विकल्प
four
three
remains same
propane
ns1-2
increases
six
propan-2-ol
(n-1)d1-10
two
decreases
Clemmensen’s
Wolff-Kishner
उत्तर
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are six and three.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Write IUPAC names of the following compounds
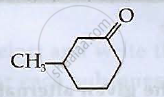
Write IUPAC names of the following compounds:
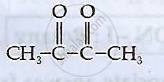
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
In which of the following compounds, the oxidation number of iodine is fractional?
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The oxidation number d-arbitral occupation and co-ordination number of Cr in the complex cis [Cr(en)2Cl2]Cl are respectively.
Ethylene diamine tetraacetate (EDTA) ion is ______.
The one that will show optical activity is: (en = ethane 1, 2-diamine)
