Advertisements
Advertisements
Question
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Options
four
three
remains same
propane
ns1-2
increases
six
propan-2-ol
(n-1)d1-10
two
decreases
Clemmensen’s
Wolff-Kishner
Solution
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are six and three.
APPEARS IN
RELATED QUESTIONS
Write structures of compounds A, B and C in of the following reactions
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
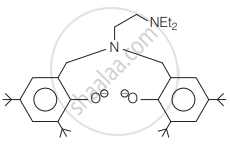
The following ligand is:
What is a chelate complex? Give one example.
Explain the following, giving two examples:
Heteroleptic
What is meant by didentate ligand?
Give two examples of ambidentate ligand.
