Advertisements
Advertisements
Question
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Solution
[Co(NH3)3Cl3] < [Co(NH3)4Cl2]Cl < [Cr(NH3)5Cl]Cl2 < [Co(NH3)6]Cl3
APPEARS IN
RELATED QUESTIONS
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Write structures of compounds A, B and C in of the following reactions
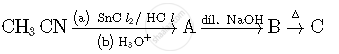
Write structures of compounds A and B of the following reaction :

According to IUPAC nomenclatures, sodium nitroprusside is named as
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
The one that will show optical activity is: (en = ethane 1, 2-diamine)
Explain the following, giving two examples:
Coordination polyhedron
Explain the following, giving two examples:
Coordination number
What is meant by didentate ligand?
