Advertisements
Advertisements
Question
Explain the following, giving two examples:
Coordination entity
Solution
A coordination entity constitutes a central metal atom or ion bonded to a fixed number of ions or molecules. For example, [CoCl3(NH3)3] is a coordination entity in which the cobalt ion is surrounded by three ammonia molecules and three chloride ions. Other examples are [Ni(CO)4], [PtCl2(NH3)2], [Fe(CN)6]4−, [Co(NH3)6]3+.
APPEARS IN
RELATED QUESTIONS
What is meant by unidentate ligand?
Amongst the following, the most stable complex is:
Write IUPAC names of the following compounds:
Write the IUPAC name of the following complex : [Co(NH3)5(CO3)]Cl.
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the structures of compounds A, B and C in the following reactions:
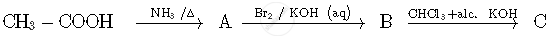
Write the structures of compounds A, B and C in the following reactions
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
Write structures of compounds A and B of the following reaction :
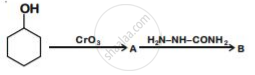
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
Which one of the following does not achieve an octet of electrons in the central atom?
Which one of the following ligands forms a chelate?
Why chelate complexes are more stable than complexes with unidentate ligands?
The following ligand is:
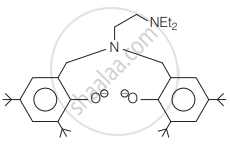
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
What is a chelate complex? Give one example.
What is meant by the chelate effect? Give an example.
Give two examples of didentate ligand.
What is meant by ambidentate ligand?
