Advertisements
Advertisements
Question
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
Options
Assertion and reason both are true, reason is correct explanation of assertion.
Assertion and reason both are true but reason is not the correct explanation of assertion.
Assertion is true, reason is false.
Assertion is false, reason is true.
Solution
Assertion and reason both are true but reason is not the correct explanation of assertion.
Explanation:
In the complexes, \[\ce{Co}\] exists as \[\ce{Co^{2+}}\] and \[\ce{Fe}\] as \[\ce{Fe^{2+}}\]. Both of the complexes becomes stable by oxidation of metal ion to \[\ce{Co^{3+}}\] and \[\ce{Fe^{3+}}\].
APPEARS IN
RELATED QUESTIONS
Write IUPAC names of the following compounds:
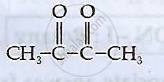
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the structures of compounds A, B and C in the following reactions:
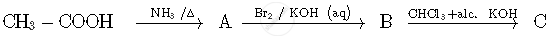
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
The most stable ion is:-
What are Homoleptic complexes?
Why chelate complexes are more stable than complexes with unidentate ligands?
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
