Advertisements
Advertisements
Question
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Solution
[Cr(NH3)3Cl3]
IUPAC name: Triamminetrichloridochromium(iii)
APPEARS IN
RELATED QUESTIONS
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
Write applications of co-ordination compounds in medicine and electroplating.
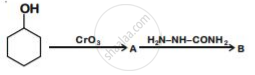
Write structures of compounds A and B of the following reaction :
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
Oxidation number of carbon in CH2Cl2 is
The most stable ion is:-
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
