Advertisements
Advertisements
Question
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
Solution
The central atom is electron pair acceptor so it is a Lewis acid.
APPEARS IN
RELATED QUESTIONS
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Write structures of compounds A, B and C in of the following reactions
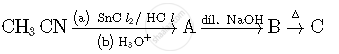
Write the IUPAC name of the following complex : [Co(NH3)5(CO3)]Cl.
Write the structures of compounds A, B and C in the following reactions
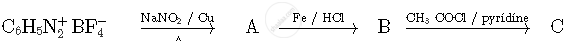
Write structures of compounds A and B of the following reaction :

Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
According to IUPAC nomenclatures, sodium nitroprusside is named as
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
Explain the following, giving two examples:
Homoleptic
Explain the following, giving two examples:
Heteroleptic
