Advertisements
Advertisements
Question
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Solution
When aqueous KCN solution is added to aqueous copper sulphate solution, [Cu(CN)4]2− coordination species is obtained as follows:
\[\ce{[Cu(H2O)4]^2+ + 4CN- -> [Cu(CN)4]^2- + 4H2O}\]
The coordination species thus formed [Cu(CN)4]2− is very stable because CN– is a strong ligand. Hence, on passing H2S gas through this solution, CuS precipitate is not obtained because free Cu2+ ions are unavailable.
APPEARS IN
RELATED QUESTIONS
What is meant by unidentate ligand?
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Write the structures of compounds A, B and C in the following reactions
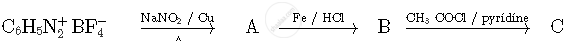
A group of atoms can function as a ligand only when:
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The coordination number of the central ion may be obtained from:
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
Which one of the following does not achieve an octet of electrons in the central atom?
Which one of the following ligands forms a chelate?
Ethylene diamine tetraacetate (EDTA) ion is ______.
Ethylene diaminetetraacetate (EDTA) ion is ______
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
The following ligand is:
Which of the following species cannot act as a ligand? Give reason.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Explain the following, giving two examples:
Ligand
Give two examples of didentate ligand.
Give two examples of ambidentate ligand.
