Advertisements
Advertisements
Question
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Solution
Oxidation state = +3,
Coordination number = 6,
3d6 configuration = \[\ce{t^6_{2g}e^0_g}\]
APPEARS IN
RELATED QUESTIONS
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write structures of compounds A, B and C in of the following reactions
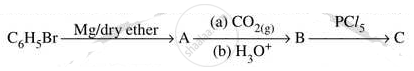
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write structures of compounds A and B of the following reaction :
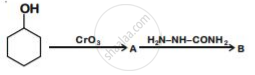
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Which of the following is non-ionizable?
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Ethylene diamine tetraacetate (EDTA) ion is ______.
The following ligand is:
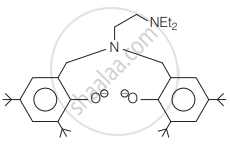
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Glycinato ligand is ______.
What is a chelate complex? Give one example.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Heteroleptic
What is meant by ambidentate ligand?
