Advertisements
Advertisements
प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
उत्तर
Oxidation state = +3,
Coordination number = 6,
3d6 configuration = \[\ce{t^6_{2g}e^0_g}\]
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write IUPAC names of the following compounds
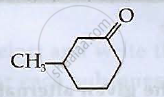
Write structures of compounds A, B and C in of the following reactions
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
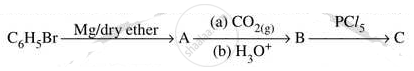
Write structures of compounds A and B of the following reaction :
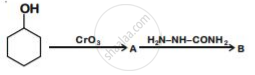
Which of the following represents a chelate ligand?
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
In which of the following compounds, the oxidation number of iodine is fractional?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Which one of the following ligands forms a chelate?
Which of the following ligands can exhibit linkage isomerism?
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
What is meant by the chelate effect? Give an example.
