Advertisements
Advertisements
प्रश्न
Write IUPAC names of the following compounds
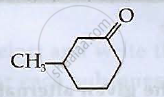
उत्तर
3-Methylcyclohexanone
APPEARS IN
संबंधित प्रश्न
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
IUPAC name of the following compound is

(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
Oxidation number of carbon in CH2Cl2 is
In which of the following compounds, the oxidation number of iodine is fractional?
According to IUPAC nomenclatures, sodium nitroprusside is named as
What are Homoleptic complexes?
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Glycinato ligand is ______.
The one that will show optical activity is: (en = ethane 1, 2-diamine)
What is a chelate complex? Give one example.
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Coordination polyhedron
Explain the following, giving two examples:
Homoleptic
Explain the following, giving two examples:
Heteroleptic
Give two examples of unidentate ligand.
What is meant by didentate ligand?
What is meant by ambidentate ligand?
