Advertisements
Advertisements
प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
उत्तर
Oxidation state = +2,
Coordination number = 4,
d7 configuration = \[\ce{t^5_{2g}e^2_g}\]
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Explain the following, giving two examples:
Coordination entity
IUPAC name of the following compound is

(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC names of the following compounds
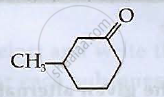
Write the structures of compounds A, B and C in the following reactions:
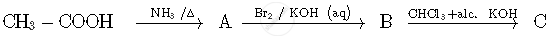
Write the structures of compounds A, B and C in the following reactions
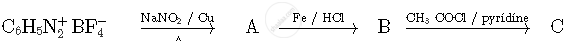
Which of the following represents a chelate ligand?
Ligand (en) is an example of ___________.
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
Which of the following ligands can exhibit linkage isomerism?
Ethylene diaminetetraacetate (EDTA) ion is ______
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
Give two examples of unidentate ligand.
Give two examples of ambidentate ligand.
