Advertisements
Advertisements
Question
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
Solution
The oxidation state of iron in this complex ion is +2.
Electronic configuration of Fe = [Ar] 3d64s2
Electronic configuration of Fe2+ = [Ar] 3d6
The iron (II) ion must provide six vacant orbitals to accommodate the six electron pairs from the six cyanide ions. This can be achieved by the hybridisation method in which the electrons of the d-subshell get paired since the CN– ions are strong field ligands.
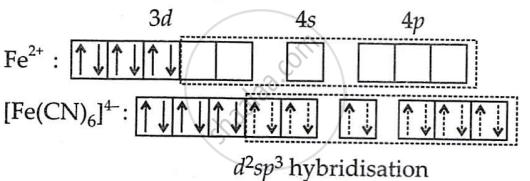
Hence, six electron pairs from six cyanide ions occupy six hybrid orbitals of iron (II) ion. Thus, no unpaired electrons are in any orbital, so [Fe(CN)6]4− shows diamagnetism. Hence, [Fe(CN)6]4− is diamagnetic and octahedral.
APPEARS IN
RELATED QUESTIONS
Explain on the basis of valence bond theory that [Ni(CN)4]2− ion with square planar structure is diamagnetic and the [NiCl4]2− ion with tetrahedral geometry is paramagnetic.
Predict the number of unpaired electrons in the square planar [Pt(CN)4]2− ion.
[Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2− is diamagnetic. Explain why?
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(C2O4)3]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Explain the geometry of `[Co(NH_3)_6]^(3+)` on the basis of hybridisation. (Z of Co = 27)
[NiCl4]2- is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why? (Atomic no. Ni = 28)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Mn(CN)6]^{3-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Cr(H2O)6]^{3+}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[FeCl6]^{4-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
The type of hybridization involved in Octahedral complexes is ______.
Write the hybridization and shape of the following complexes:
[Ni(CN)4]2−
Which of the statement given below is incorrect about H2O2?
As the s-character of hybridised orbital increases, the bond angle
Using valence bond theory, predict the hybridization and magnetic character of the following:
[CoF6]3– [Atomic number of Co = 27]
The magnetic moment of [NiCl4]2− is ______.
[Atomic number: Ni = 28]
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
During chemistry class, a teacher wrote \[\ce{[Ni(CN)4]^2-}\] as a coordination complex ion on the board. The students were asked to find out the magnetic behaviour and shape of the complex. Pari, a student, wrote the answer paramagnetic and tetrahedral whereas another student Suhail wrote diamagnetic and square planer.
Evaluate Pari’s and Suhail’s responses.
