Advertisements
Advertisements
प्रश्न
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
विकल्प
A - (1), B - (2), C - (4), D - (5)
A - (4), B - (3), C - (2), D - (1)
A - (3), B - (2), C - (4), D - (1)
A - (4), B - (1), C - (2), D - (3)
उत्तर
A - (4), B - (3), C - (2), D - (1)
Explanation:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 4. Yellowish orange |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 3. Pale blue |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 2. Green |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 1. Violet |
APPEARS IN
संबंधित प्रश्न
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
How is Benzonitrile converted to Benzophenone?
Write the structures of compounds A, B and C in the following reactions:
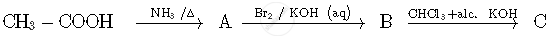
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
Which of the following represents a chelate ligand?
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
The most stable ion is:-
Which of the following ligands can exhibit linkage isomerism?
Which of the following species cannot act as a ligand? Give reason.
What is meant by ambidentate ligand?
