Advertisements
Advertisements
प्रश्न
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
विकल्प
A - (5), B - (4), C - (1), D - (2)
A - (3), B - (4), C - (5), D - (1)
A - (4), B - (3), C - (2), D - (1)
A - (3), B - (4), C - (1), D - (2)
उत्तर
A - (5), B - (4), C - (1), D - (2)
Explanation:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 5. magnesium |
| B. Blood pigment | 4. iron |
| C. Wilkinson catalyst | 1. rhodium |
| D. Vitamin B12 | 2. cobalt |
APPEARS IN
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Write structures of compounds A, B and C in of the following reactions
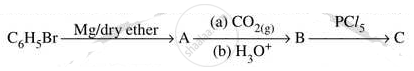
Write the structures of compounds A, B and C in the following reactions:
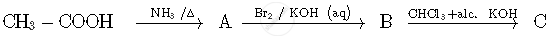
Write structures of compounds A and B of the following reaction :

Write structures of compounds A and B of the following reaction :
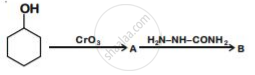
Which of the following represents a chelate ligand?
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
