Advertisements
Advertisements
प्रश्न
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
विकल्प
A - (1), B - (2), C - (4), D - (5)
A - (4), B - (3), C - (2), D - (1)
A - (5), B - (1), C - (4), D - (2)
A - (4), B - (1), C - (2), D - (3)
उत्तर
A - (1), B - (2), C - (4), D - (5)
Explanation:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 4. + 2 |
| D. \[\ce{[Co2(CO)8]}\] | 5. + 3 |
APPEARS IN
संबंधित प्रश्न
What is meant by unidentate ligand?
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the structures of compounds A, B and C in the following reactions
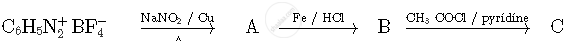
Which of the following represents a chelate ligand?
A group of atoms can function as a ligand only when:
The coordination number of the central ion may be obtained from:
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
According to IUPAC nomenclatures, sodium nitroprusside is named as
