Advertisements
Advertisements
प्रश्न
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
उत्तर
Copper ferrocyanide
APPEARS IN
संबंधित प्रश्न
Write applications of co-ordination compounds in medicine and electroplating.
Write structures of compounds A, B and C in of the following reactions
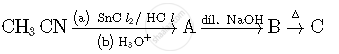
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
Which of the following represents a chelate ligand?
Ligand (en) is an example of ___________.
In which of the following compounds the oxidation state of the nickel atom is 0?
Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Which one of the following does not achieve an octet of electrons in the central atom?
The nature of hybridisation in the ammonia molecule is
The oxidation number d-arbitral occupation and co-ordination number of Cr in the complex cis [Cr(en)2Cl2]Cl are respectively.
According to IUPAC nomenclatures, sodium nitroprusside is named as
Which of the following ligands can exhibit linkage isomerism?
Ethylene diamine tetraacetate (EDTA) ion is ______.
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
Glycinato ligand is ______.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Explain the following, giving two examples:
Coordination polyhedron
Explain the following, giving two examples:
Coordination number
Give two examples of unidentate ligand.
Give two examples of ambidentate ligand.
