Advertisements
Advertisements
Question
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Options
\[\ce{Cu^{2+} + 4NH3 ⇌ [Cu(NH3)4]^{2+}, logK = 11.6}\]
\[\ce{Cu^{2+} + 4CN^- ⇌ [Cu(CN)4]^{2-}, logK = 27.3}\]
\[\ce{Cu^{2+} + 2en ⇌ [Cu(en)2]^{2+}, logK = 15.4}\]
\[\ce{Cu^{2+} + 4H2O ⇌ [Cu(H2O)4]^{2+}, logK = 8.9}\]
Solution
\[\ce{Cu^{2+} + 4CN^- ⇌ [Cu(CN)4]^{2-}, logK = 27.3}\]
Explanation:
The greater the value of log K, the greater will be stability of complex compound formed.
For reaction,
\[\ce{Cu^{2+} + 4CN^{-} -> [Cu(CN)4]^{2-}}\]
`"K" = (["Cu"(("CN")_4)^(2-)])/(["Cu"^(2+)]["CN"^(-)]^4)` and log K = 27.3
For this reaction, log K has highest value among the given four reactions. Hence, K will also be higher among these four, i.e., stability of the complexes will be highest among these four complexes.
APPEARS IN
RELATED QUESTIONS
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Write structures of compounds A, B and C in of the following reactions
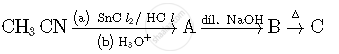
Write structures of compounds A and B of the following reaction :
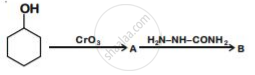
Ligand (en) is an example of ___________.
The most stable ion is:-
According to IUPAC nomenclatures, sodium nitroprusside is named as
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
What is a chelate complex? Give one example.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
